A powder that seems dry but it contains almost only waterfall: Dry water arises from a physical transformation that completely changes the way the liquid behaves. It is not a new substance, but a new way of organizing water, divided into microdrops and covered by solid particles. Understanding how this structure is formed requires entering into the microscopic mechanisms that make it stable, while experiments show how it manages to modify fundamental properties, such as smoothness of materials or interaction with gases. It is precisely from these results that the most interesting applications emerge, ranging from the engineering of construction materials to technologies for storage and recycling gas capture, like carbon dioxide.
How dry water is formed: the composition
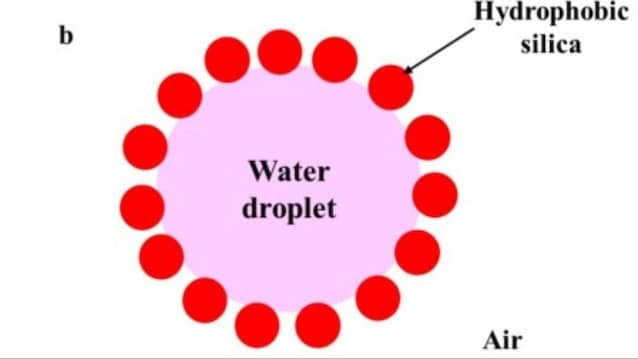
Dry water is a powder composed largely of water, up to 95–98% by weight, but which behaves like a solid material. Basically it’s not the water that changes state, but something more interesting happens. During preparation, the water is agitated with speeds up to tens of thousands of revolutions per minute, together with particles of silica very small and hydrophobic (i.e. they “repel” water). This process is conducted at high energy and breaks the liquid into microscopic dropletseach of which is covered by a stable shell of solid particles.
The result is a structure with water inside and a “network” of silica nanoparticles. This coating prevents the drops from merging together (coalescence) and therefore to return to liquid water. The stability of the silica shell depends on several factors: particle size, mixing energy, affinity between water and solid surface (contact angle). If the silica is not hydrophobic enough, the system does not work: instead of powder you get a suspension or mousse.
What changes compared to normal water
The key point about dry water versus liquid water is that dry water is no longer continuous, but divided into millions of isolated micro-drops. And this completely changes the behavior of the system. A very clear example comes from studies of granular materials: when normal water is added to sand, they form liquid bridges between the granules that they make it compact and not very smooth.
In fact, the sliding angle goes from approximately 38.7° to 56.2° already with only 0.5% of water. With dry water, however, the opposite happens: the water remains “trapped”, no liquid bridges are formed and therefore the material continues to behave like a fluid powder. This is one of the most interesting results from an application point of view.
Experiment and main results
One of the most concrete studies is that conducted by Leigh Duncan Hamilton and colleagues atInstitute for Particle Technologywho tested dry water mixed with sand. The goal was to insert water into a granular material without ruining its properties.
They prepared several variations of dry water with different water/silica ratios. Then, they mixed them with sand (particles ~1 mm) and finally varied the water content up to 5 wt%. They observed that water can be incorporated without loss of smoothness; that the presence of silica prevents the formation of liquid bridges and that even small amounts of silica are sufficient to stabilize the system.
Dry water, however, is not stable forever and although it seems like a “perfect” material, it has limitations: can break under mechanical stressthe drops can gather over time, the reusability it’s still an open problem.
The various applications of powdered water
The interest in dry water arises precisely from what it makes possible in many fields, from construction to cosmetics.
1. Materials and constructions
In construction, dry water can be used to store water in materials, improve processes such as 3D printing concrete, compensate for water losses and cool the systems.
2. Gases and the environment
According to a more recent review, dry water greatly increases the contact between water and gas. This has a direct effect on gas hydrate formation, capture of carbon dioxide (CO2), storage of methane or hydrogen. The classic problem is that gases dissolve little in water, but here each drop is an available surface and therefore the process accelerates.
3. Pharmaceuticals and cosmetics
However, an important use in pharmaceuticals and cosmetics already emerges from the initial studies. An important mechanism in this area is the ability to transport active substances in a protected form and release them only when needed. This is because water can be released simply by mechanical stress (for example rubbing).
