We find many oils on the market: peanuts, seeds, olive oil itself, but to understand which is really the best oil for frying, it is necessary analyze its chemical composition. Spoiler: the podium belongs to olive and peanut oil. During frying, the oil transfers heat to the food, but not all oils behave in the same way when we bring them to high temperatures: some resist better, others degrade more quickly, also producing unwanted substances. Oils are mainly composed of triglyceridesmade up of saturated or unsaturated fatty acids. The double bonds (unsaturations) of the latter, are vulnerable points which at high temperatures react with environmental oxygen in processes “thermooxidation“, forming even unwanted substances such asacrolein (classified by International Agency of Research on Cancer, IARC as probably carcinogenic). The richer an oil is in these unsaturations, the more easily it degrades during frying. Choosing only oils with saturated fats is not the best solution, as they are unhealthy. The right compromise is an oil with few unsaturations, which is stable during heating, but also nutritionally valid. This is the case ofolive oil and dell‘peanut seed oilwhose structures and compositions allow it to resist heat better than, for example,sunflower seed oil.
Frying oils from a chemical point of view
Edible oils are mixtures of lipidsthat is, fat, like sterols (whose parent is cholesterol), terpene lipids (such as vitamin E or squalene) and carotenoids. However, the main component is represented by triglycerideswhich can constitute up to 98% of total fats. Triglycerides are molecules formed by a basic structure, the glycerine (or glycerol), to which three molecules of fatty acidsthat is, long chains made up of carbon and hydrogen, divided into two main categories:
- Fatty acids saturated: no double bonds
- Fatty acids unsaturated: they have double bonds, also called unsaturations, between the carbon atoms in their structure. If they have only one unsaturation, we talk about fatty acids monounsaturated (MUFA); if they have more unsaturations along the chain we talk about fatty acids polyunsaturated (PUFA).
Precisely these double bonds represent the reactive centers of the molecule, which can change during chemical reactions. In fact, while we fry, many reactions can occur, including those of thermoxidation. This term indicates the set of oxidation reactions that occur in fats when they are exposed to high temperatures and environmental oxygen, such as when we heat oil in a pan while frying.
During these processes, unsaturated fatty acids react with oxygen (O2), forming so-called ROS, reactive oxygen species, and oxidized compounds: some are volatile molecules, that is, they easily pass into the air and therefore move away through the vapor, while other non-volatile molecules they remain in the oil and can be transmitted to food. An important example is theacroleinclassified byIARC in group 2A, i.e. that of substances probably carcinogenic to humans.
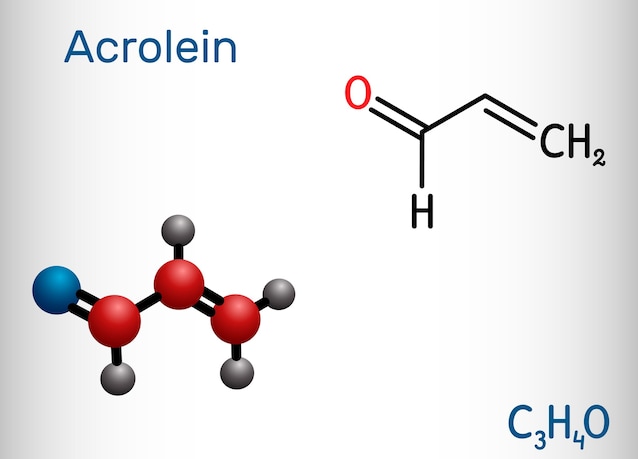
Olive and peanut oil, the compromise between unsaturations and stability: scientific studies
In general, the more an oil has fatty acids with unsaturations, the more sensitive it will be to thermooxidation reactions and therefore to degradation. One might therefore think that the best oils for frying are those composed exclusively of saturated fatty acids, as they have no double bonds and are therefore more stable at high temperatures. However, this conclusion is misleading: oils consisting only of saturated fatty acids are very stable, but they are not a good choice from a nutritional point of view and can increase blood cholesterol values, as also reported by many articles, for example a study published in Lipids in 2010.
Consequently, the best oils for frying are actually those that possess unsaturated fatty acids, but with few total unsaturations: the perfect compromise is represented by oils with a high MUFA content and a low PUFA content, ideally below 3%. What edible oils meet these requirements? First of all there is theolive oilthe fourth most used in Europe and rich in MUFA, especially oleic acidwhich gives it a high oxidative stability. Then there is thepeanut oilextracted from the seeds ofArachis hypogaeaalso with a high content of monounsaturated fatty acids.
On the contrary, thesunflower seed oil it is rich in PUFA, especially of linoleic acidwith two unsaturations. The difference may seem small, but not on a large scale. To give a basic example, let’s imagine a triglyceride with three molecules of oleic acid (monounsaturated, i.e. only one double bond that can be “attached”): we have three reactive centers which can undergo thermooxidation. If instead we have a triglyceride with three molecules of linoleic acid (which has two double bonds), the quantity of sensitive reactive centers double at six.
It is no coincidence that there is also thehigh oleic sunflower oil: this is a variant obtained by selecting seeds with more oleic acid and less unstable PUFA. In this way we obtain an oil that is more resistant to heat and therefore more suitable for frying than traditional sunflower seed oil.
Another point in favor of oils with low PUFA content, such as olive oil and peanut oil, is the presence of a high smoke pointi.e. the temperature at which the oil begins to decompose producing potentially harmful compounds. Research published in Foods in 2024 showed, for example, that sunflower oil it degrades about twice as fast compared to olive oil when frying. In other words, olive oil is great for frying as it can be heated to high temperatures without degrading, or at least degrading more slowly than other oils.
