Why in winter the surface water of rivers, pools and lakes can freeze but The sea does not ice? The answer lies in its salinity, that is, the content of salt dissolved in the sea, which is quite high (approximately 35 grams each liter) to lower the freezing temperature to -2 ° C. It is the same principle that winter throws the salt on the roads, to prevent them from gnoot when temperatures go down below zero. To prevent the sea ice from the sea also contribute the Continuous remediation of water masses caused by the currents, which maintain the Taverage hemperature at about 3.5 ° Cand the gigantic volume of the sea and the oceans. Actually Even the icing oceanbut only at temperatures constantly low of Arctic and Antarctic, where it can reach an area of 18.5 million km², while it is liquid to wet our coasts.
The sea does not freeze because of the salt
We all know: the water ice icges at 0 ° C and bubbles at 100 ° C. Right, right? Well, not really. Only pure water, that without dissolved saltsrespect these conditions. Did they always tell us wrong so? Let’s say no, why The variation is so small That we can approach by saying that “the water boils at one hundred degrees and freezes at zero degrees”.
For sea waterhowever, the speech is a little different, since the amount of salt It is conspicuous. The ocean contains, in fact, approximately 35 grams of rooms per liter which corresponds to a Average salinity of 35 ‰ (let’s say average because not all points of the ocean have the same salinity). The presence of dissolved salts generates an effect called cryoscopic lowering: lowers the freezing point of water to approx –2 ° C (28.4 ° F), compared to the classic 0 ° C of the “sweet” water, preventing the marine surface from freezing to typical terrestrial temperatures.
The effect of marine currents
There temperature plays a fundamental role in stratification and in remediation of large masses of water. When the sea surface cools down and approaches the passage of state, the salted water becomes more densesinks and “exchanges of place” with the hot water that is deeper. The continuous movement of currents and water masses generates a thermal refurbishment that prevents the surface of the sea, in most cases, to reach very low temperatures by slowing down its freezing.
Also i winds They can affect this phenomenon, moving the water masses and promoting the mixing effect. On average we can say that the Ocean temperature is around the 3.5 ° C (38.3 f). On a global level, the remission of the masses of water based on density and temperature is called “terrestrial thermoaline circulation” and one of the most important cold currents for this engine is the North Atlantic Deep Water (NADW).
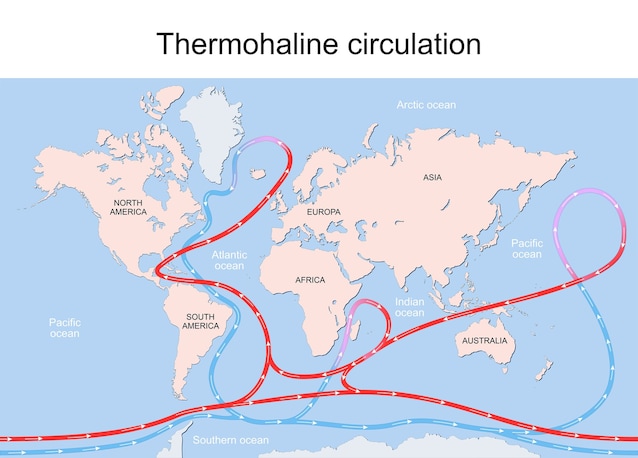
The effect of the volume of the oceans on freezing
The ocean is giganticcontains enormous volumes of water that have their own Thermal capacity: the greater the volume of water to cool, greater energy It must be removed to favor their freezing. Just to be clear, it’s like trying to freeze in the freezer, with the same times, a full glass or a bottle of water: perhaps after an hour the water in the glass will be frozen, but certainly not a whole liter.
Unless we color that liter of water in a tray … at that point we will have increased the surface of water exposed to freezing (in technical terms we have increased the surface/volume ratio) And therefore it could freeze before.
In the sea, however, the surface/volume ratio is very low Since the seas are very deep, therefore to encourage freezing, the thermal exchange surface should be hypothetically increasing in order to more easily disperse the heat held by the mass of water. In short, being very wide and deep The oceans tend to remain liquid As we know them.
Marine ice: when it is formed and because it is sweet
Despite what has just been said, sometimes, Even the ocean can freeze Originning the sea ice. It is formed in the polar regions where surface temperatures are sufficiently low and constant to allow salted water to form a stable crystalline lattice. In these areas, irradiation (i.e. the transfer of energy from one body to another through electromagnetic waves) is less powerful and the albedo effect, i.e. the ability of a surface (in our case the ocean) to reflect solar radiation, favors a less absorption of sunlight. Marine ice originates, grows and always merges into the ocean where, however, it can be covered by abundant snow.

During the passage of status from solid liquid, a rather curious thing takes place: Only water molecules form an elegant and compact crystalline structurewhile the ions remain dissolved in liquid water all around. The salt is then excludedexpelled from the crystallization process thus making the “sweet” sea ice and consequently increasing the salinity of the underlying water. The ice that forms for this effect of Rojectton Brine it is not completely without salt, but it contains very little, so much so that it can say that it is almost fresh water (older is, the less salt contains).
The difference between the ice of the Arctic and the Antarctica
The type of sea ice depends a lot on the geographic region in which it is formed and is very different between Arctic and Antarctica both as regards Seasonal extension and contractionthat for it thickness and the interaction with winds and currents. It tends to be the Arctic one is more often and lasting, less mobile, with a lower and asymmetrical extension. In the’Arctic Marine ice is very common and can cover up to about 15.5 million km² in winter, in Antarcticaon the other hand, up to about 18.5 million km². It can form more or less thick, wide or elongated layers, helped by the force of the currents.

Let us remember that Antarctica is a real continent with different thermal dynamics compared to the polar flavor polar cap. In the southern hemisphere they are mostly ice seasonal which blends almost completely in summer, in the Arctic instead a part persists and becomes multi -year ice. These and other characteristics are studied by scientists, climatologists and ecologists who constantly monitor ice from all over the world studying their dynamics and creating statistical models from year to year.
Difference between sea ice and earthly ice
There are actually differences between sea ice and terrestrial ice that mainly concern theirs origin. Sea ice is formed from freezing of surface salted waterfloats directly on the sea, can vary extension quickly and be very ephemeral.
The Earth’s ice that reaches the sea Instead, it comes from the accumulation of snow and ice on earth, (therefore from fresh meteoric water) and tends to be very compact for an effect of slow compression of the snow and the mass itself. It can be in the form of glaciers, caps or reach the sea and detach yourself in large blocks called iceberg.
