Five children between 5 and 11 years old were hospitalized at the Umberto I Polyclinic in Rome after a bath in the public swimming pool of a sports center on Sunday, with respiratory symptoms And skin irritation possibly due to chlorine poisoningwhich has an antimicrobial action and is used in disinfectant compounds in swimming pools. The child in more worrying conditions ended up in intensive care with serious breathing difficulties: at the moment he is kept in a pharmacological coma and would risk irreversible neurological damage. The plant was seized and the Rome prosecutor’s office launched a survey on the accident, which could be due to a pressure drop which would have caused accumulations of chlorine then downloaded to too high concentrations in the area where children were located.
According to the first reconstructions, one of the children would report to the mother that The water of the swimming pool was becoming yellow: if confirmed, this could indicate that in the water of the swimming pool it has freed itself gaseous chlorine (Cl2), a toxic substance that can cause serious effects to the respiratory tract, the mucous membranes and the skin.
Disinfectant action and safety
Chlorine in swimming pools is used as a disinfectant because of its effective antimicrobial action. For this purpose different compounds are used with disinfectant action, depending on the size of the swimming pool and other parameters.
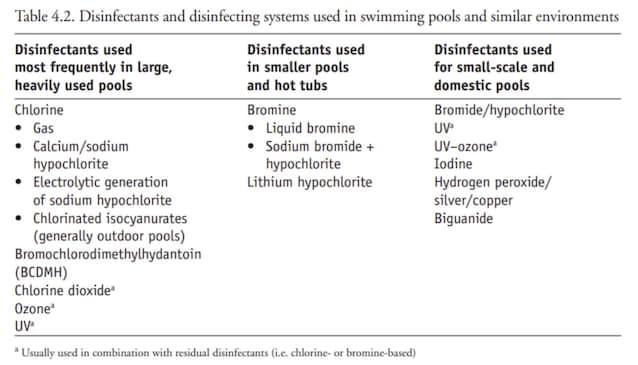
However, having chlorine an irritating action also for the human organism, they exist Specific guidelines which establish the maximum eligible concentrations in a swimming pool to avoid adverse effects. The WHO recommends not to exceed 3 milligrams per liter for public and semi-public swimming pools; In Italy the concentrations are usually stood between 0.5 and 1.5 milligrams per liter.
By maintaining the concentrations of these compounds within the established limits, you can enjoy a day in the pool In total safety.
The ideal values of chlorine pH in swimming pools: possible risks
However, the question does not close here: some of the disinfectants used in the swimming pools are in fact acids or basictherefore able to alter the water pH. Always according to WHO indications, the pH must be maintained Between 7.2 and 7.8.
A common disinfectant is the sodium hypochlorite (Naclo), which is basic and therefore tends to raise the pH of the water. Its use therefore requires theAdding acids To lower the pH and bring it back to the optimal interval. Typical correctors in this case can be the hydrogen sodium sulphate (Nahso4) or an watery solution of hydrochloric acid (HCL).
In the latter case – or in cases where a disinfectant is used that free sodium hypochlorite – however, a problem may arise if for any reason the instructions are not followed by letter: the hypochlorite sodium can in fact react with hydrochloric acid to form (in addition to sodium chloride and water) gaseous chlorinewhich if inhaled in sufficient quantities can be very toxic and cause serious irritations to the respiratory tract, skin and mucous membranes. This is the chemical reaction:
Naclo + 2hcl → Cl2 + Nacl + h2OR
This, among other things, is the reason why we must not mix bleach and muriatic acid. Also diluted in the water, gaseous chlorine can give water one yellowish coloringas it seems to have happened in the Roman swimming pool where the accident occurred.
In conclusion, The pools are safe When the levels of chlorine and pH are maintained within optimal ranges: for this reason it is important for the operators of the systems follow precisely to national and regional regulations.
