By often washing glasses in the dishwasher, they can undergo a process of “opacification”, i.e. the loss of transparency following the formation of white marks. It is no coincidence that in English this phenomenon is called cloudinessfrom cloud (“cloud”): the glass loses its clarity and takes on an appearance milkyjust as if a cloud had been trapped inside the glass.
At first glance you might think it’s simple glass foggingdue to contact between a cold surface and hot, humid air: the water vapor present in the dishwasher seems in fact the most intuitive culprit. In that case, however, it would be a temporary effect destined to disappear once the condensation has evaporated. Instead, it is often a process irreversible, caused by a series of chemical reactions that take place right on the surface of the glass. High heat and alkaline cleaners they slowly attack the structure of the glassmaterial composed mainly of silicacreating microscopic irregularities. These alterations make the surface less uniform and change the way in which light passes through it: instead of proceeding linearly, it is diffused in multiple directions, creating that typical opaque and milky effect. In many cases, too limestone deposits and other substances present in hard water contribute to further accentuating this phenomenon.
How water and heat corrode glass
Glass is a amorphous solidthat is, a material which, although rigid, does not have an ordered crystalline structure. Its main component is the silicon dioxide (SiO2), also known as silica. To be more precise, glass is made up of a three-dimensional network where silica repeats numerous times: each silicon atom is bonded to four oxygen atoms, forming a continuous and compact structure.
According to research published by the University of Bayreuth, repeated exposure of glass to high temperatures and aggressive alkaline detergents during washing in the dishwasher it can induce phenomena of corrosion. By glass corrosion we mean a set of irreversible chemical-physical processes in which the surface of the material reacts with the surrounding environment, determining the progressive dissolution of its surface structure. These phenomena they alter the microstructure of the glasscausing permanent morphological changes that increase surface roughness and the formation of tiny cavities. At a macroscopic level, these alterations can manifest themselves as a loss of brilliance, opacification and other visual defects.
Why does it appear opaque? Question of light and “micro-scratches”
A perfectly transparent glass appears as such because its structure is uniform: the light that passes through it encounters a homogeneous material and can propagate in a relatively orderly way. However, when the glass is progressively ruined by washing in the dishwasher, this microscopic uniformity disappears. As already seen, the warm, humid and highly alkaline (i.e. basic) environment favors chemical corrosion of the surface layer of the glass, modifying the surface which will go from smooth and compact to rough with porosity and areas altered from a chemical-physical point of view.
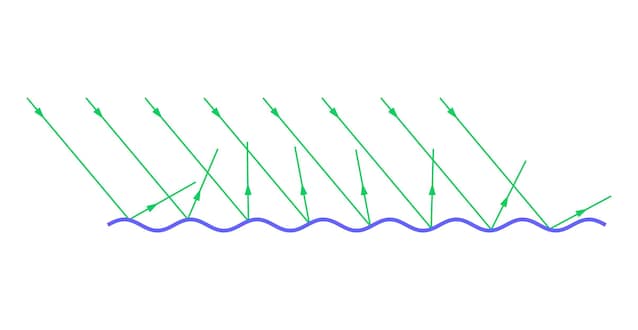
And this is where physics comes in. When light encounters a perfectly regular surface, it continues its path in a more or less coherent way. If instead he meets microscopic inhomogeneities – cavities, roughness, local variations in chemical composition – no longer passes through the material in an orderly line, but comes deflected and reflected in many directions. This phenomenon is called diffusion of light (light scattering), and is the main responsible for the opaque effect: the light, “scattering” in all directions, does not allow us to clearly see what is on the other side of the glass, but on the contrary the eye perceives that typical milky halo.
In the case of glasses washed in the dishwasher, the phenomenon can be amplified by other secondary mechanisms, such as the formation of surface layers due to the deposition of carbonates (such as limestone) and other residues deriving from particularly hard water, i.e. with high concentrations of mineral ions.
