Silica gel is that material in granules that we find, among other things, in the famous “DO NOT EAT” sachets, often stuck in shoe boxes and in packaging in general. Its function is as “simple” as it is important: absorb moisture. Precisely because of this dehydrating capacity it is also used in other areas, where it is necessary to keep something dry, be it electronic components, books or indeed clothes and shoes. But how does it “capture” the water present in the air so well? The answer lies in his chemical structure. Silica gel is nothing but amorphous silica which is arranged in a real porous network. It’s like it’s one tiny network full of holes (the pores) arranged here and there ready for capture water moleculesequally small. In addition to the presence of pores, thanks to some chemical groups capable of binding waterthe same gel surface helps absorb humidity. In this way the silica gel is able to retain large quantities of H2Or on its surface without becoming soft or melting. Basically, his tricks are: lots of pores, lots of surface area and lots of places where water sticks. In this way, it manages to retain up to 40% of its weight. It is a simple, stable and effective mechanism, which makes silica gel one of the best technical desiccants around.
What silica gel is and how it works: a giant hidden surface
At first glance it looks like sand, but from a chemical point of view, silica gel is amorphous silicon dioxidea non-crystalline silica structure, i.e. not ordered and regular in space, but messy and “chaotic”. This disorder is precisely what allows us to have a porous structure capable of eliminating humidity from the air; to obtain it, particular chemical procedures called “sol–gel” These processes create an irregular three-dimensional network with microporesless than 2 nanometers in size, e mesoporesup to 50 nanometers in size. It is precisely this multitude of irregular cavities that makes the material so effective: a gram of silica gel can have even 700 m² of internal surfacealmost as much as two basketball courts squeezed into a teaspoon! Isn’t that impressive?
This enormous surface is the real “secret weapon”: more surface area means more space to accommodate water molecules. Thanks to the disordered architecture of amorphous silica, water can get everywhere and find a point that holds it. It’s a bit like entering a house with a thousand shelves: the more shelves you have, the more things you can place on them. Here every shelf is a pore.
Why water sticks: the chemistry of adsorption
Now let’s get to the heart of the phenomenon: adsorption. And no, we didn’t spell it wrong: we’re talking about adsorption and not absorption (like a sponge that incorporates water). It is a real chemical-physical phenomenon that we could define as a sort of “surface adhesion”. Simply put, when a gas reaches the surface of a solid material, the gas molecules – of H2Or present in the air as water vapor in our case – they can “rest” on the surface layer of the material.

Absorption (right, in orange): molecules are incorporated into the material, like a sponge absorbs water
This phenomenon can be more or less strong based on the interactions that form between the solid material and the gas it comes into contact with: for example, gaseous molecules that adsorb without particular interactions with the material can easily “desorb” and fly away again; if they are formed chemical bondsit will be very difficult, if not impossible, to break the bond between the material and the gas.
In the case of silica gel, adsorption occurs thanks to the interaction of some chemical groups present on the lattice and the water present in the air. The surface of the gel, in fact, is full of groups called “silanols” (Si-OH), formed by a silicon atom and a hydroxyl group (OH) which have a strong affinity with H molecules2O. The presence of these surface groups allows the formation of a hydrogen bonding which no longer lets the water escape. Although they are not as strong as the covalent bonds created in an actual chemical reaction, the interaction between the two groups is still strong enough to remove moisture from the air and never let it go away (unless external factors such as heat intervene).
Combined together, the formation of surface bonds and the high porous surface explain why silica gel can retain up to 40% of its weight in watera remarkable achievement for such a simple and stable material.
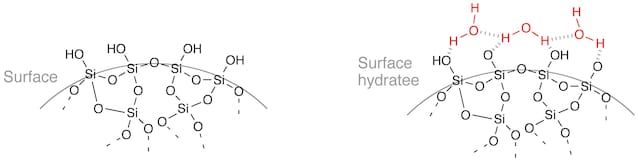
Credit: Stéphane Mons, CC BY–SA 3.0, via Wikimedia Commons
What happens next?
The interesting and curious thing is that, once saturated (i.e. when the surface has filled with water), the silica gel does not “sweat” water and does not deform by softening. It remains solid, rigid and safe even at full load. This is one of the reasons why it is chosen for delicate products such as medications, electronic devices, camera lenses, dried foods, and even museum collections.
After saturation the gel can be simply regenerated heating it. The heat breaks the hydrogen bonds with the water allowing it to evaporate; once cooled, the gel comes back ready to use again its network of pores.
In practice, silica gel works as a silent guardian against humidity: it does not make noise, it does not move, it does not change shape, but it continues to capture water indefinitely like he had a superpower. And all this thanks to structures invisible to the naked eye, pores and bonds that “trap” annoying humidity.
Sources
The Chemistry of Silica. Solubility, Polymerization, Colloid and Surface Properties, and Biochemistry. Von RK Iler. John Wiley and Sons, Chichester 1979. XXIV Brinker, C.J., & Scherer, G.W. Sol-Gel Science: The Physics and Chemistry of Sol-Gel Processing. Academic Press, 1990. The surface chemistry of amorphous silica. Zhuravlev model Chua, H., Ng, K. C., Chakraborty, A., Oo, N. M., & Othman, M. A. (2002). Adsorption characteristics of silica gel + water systems. Journal of Chemical and Engineering Data, 47, 1177-1181