The fluorinepresent in many toothpastes in concentrations of a few hundred parts per million, protects the enamel of our teeth making it more resistant to attacks by acidic compoundsthus preventing tooth decay. These are formed, for example, when bacteria normally present in the oral cavity (such as Streptococcus mutans) metabolize the sugars we ingest in the diet, transforming them into acids that attack dental enamel, the hardest tissue in the human body, leading over time to the formation of cavities. That’s why as children we were often told “Don’t eat too much candy, you’ll get cavities!” Enamel is made up of hydroxyapatitea mineral that gives teeth hardness, but is vulnerable to acidic environments: when the pH of the mouth drops, the hydroxyapatite crystals dissolve, weakening the tooth structure. Fluoride in toothpastes replaces the hydroxide ions of hydroxyapatite and forms fluorapatitemore resistant to acids. Chemically, fluorine looks like fluoride ion (F⁻)which in toothpastes is combined with a cation (a positively charged ion): among the most effective fluorinated compounds is Stannous fluoride (SnF₂).
What is hydroxyapatite, the “guardian” of our teeth
The outside of our teeth (and bones too, actually) is made up of a mineral called hydroxyapatiteconsisting mainly of calcium phosphate. It is a hierarchical and compact crystalline structure, which gives teeth their extraordinary hardness and resistance. Its chemical formula is (Ca10 (BIT4)6(OH)2) and its structure is a crystalline lattice made up of calcium (Ca), phosphate (PO43-) And hydroxide ions (OH–).

However, despite its high mechanical resistance – essential to resist the pressure of chewing – hydroxyapatite is vulnerable to acids produced by plaque bacteria. When the pH of the oral cavity is lowered (i.e. increases acidity), the hydroxyapatite crystals begin to dissolve, starting the demineralization process that weakens the tooth structure. This is where fluoride-based toothpastes come into play.
The fluoride in toothpastes serves to make the enamel more resistant
Fluoride is a chemical element that plays an important role in prevention of dental caries. For decades it has been known for its effectiveness, so much so that in many countries it can be added to drinking water, within the limits established by law (for example, in Italy the concentration of fluoride in drinking water must not exceed the concentration of 1.5 mg/L).
It is not surprising, therefore, that many toothpastes on the market contain this chemical element: fluoride can replace hydroxide ions of hydroxyapatite, forming the fluorapatitea mineral again more resistant and less susceptible to acid attacks, thus contributing to improve dental health preventing the formation of cavities.
Fluorine is part of the group of halogens (seventh group of the periodic table) and tends to acquire an electron forming an anion, i.e. an atom with a negative charge, known as fluoride ion (F–). This ion cannot be included in the formulation of toothpastes as such, but first needs to be combined with a cation (positively charged ion) in order to counterbalance the charge and obtain a neutral compound. The most used compounds are sodium fluoride (NaF), sodium monofluorophosphate (Na2BIT3F) and centous fluoride (SnF2) According to an article published on Journal of Functional Biometricsit has been shown that the Stannous fluoride it is among the most effective of all the other fluoride additives used in toothpastes: it is able to reduce the formation of bacterial plaque and counteract the demineralization of the enamel, preventing possible related disorders, such as the formation of cavities or dental sensitivity.
How acidic compounds are formed: the role of bacteria and sugars
The oral cavity hosts a vast community of microorganisms which together form the so-called oral microbiome. Our teeth are covered with an invisible film of bacteria, called biofilm.
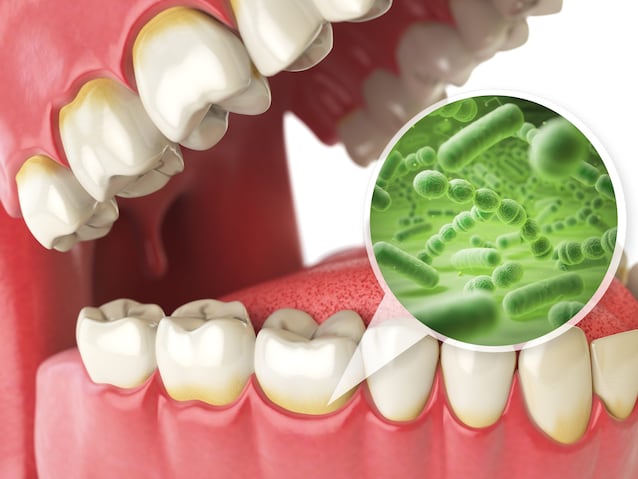
Among the main protagonists of this “microbial layer” we find the Steptococcus Mutansa bacterium capable of metabolizing fermentable carbohydrates transforming them into organic acids, such as lactic acid, acetic acid or propionic acid. But what exactly are fermentable carbohydrates? It’s about sugars such as sucrose or glucose: the more sugars we introduce through our diet (through sweets, sweets, sugary drinks), the more residues these sugars remain deposited on our teeth and can be transformed into acids by bacteria such as S. Mutans.
These acids promote demineralization of the tooth, that is, that process through which it enamel – the hard and resistant outer layer – progressively loses the minerals that compose it, such as calcium and phosphorus. Over time, the enamel can weaken to the point of forming a real cavity which, if left untreated, reaches the dentin and then the dental pulp, causing pain and infections. This is how one is born caries.
